How Mitochondrial Dysfunction Drives Chronic Fatigue
How Mitochondrial Dysfunction Drives Chronic Fatigue
If you've ever felt a bone-deep exhaustion that no amount of sleep seems to fix — an exhaustion that settles into your muscles, clouds your thinking, and makes even simple tasks feel monumental — you're not alone. For an estimated 17 to 24 million people worldwide living with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), this is daily life. And mounting evidence suggests that at the cellular level, the answer may lie in malfunctioning mitochondria.
For decades, chronic fatigue was dismissed as psychosomatic — a condition of the mind, not the body. That narrative is changing. Researchers are now documenting specific, measurable defects in mitochondrial function in ME/CFS patients, providing the kind of biological evidence that transforms a mystery into a mechanism.
The Energy Crisis: Why Your Cells Can't Keep Up
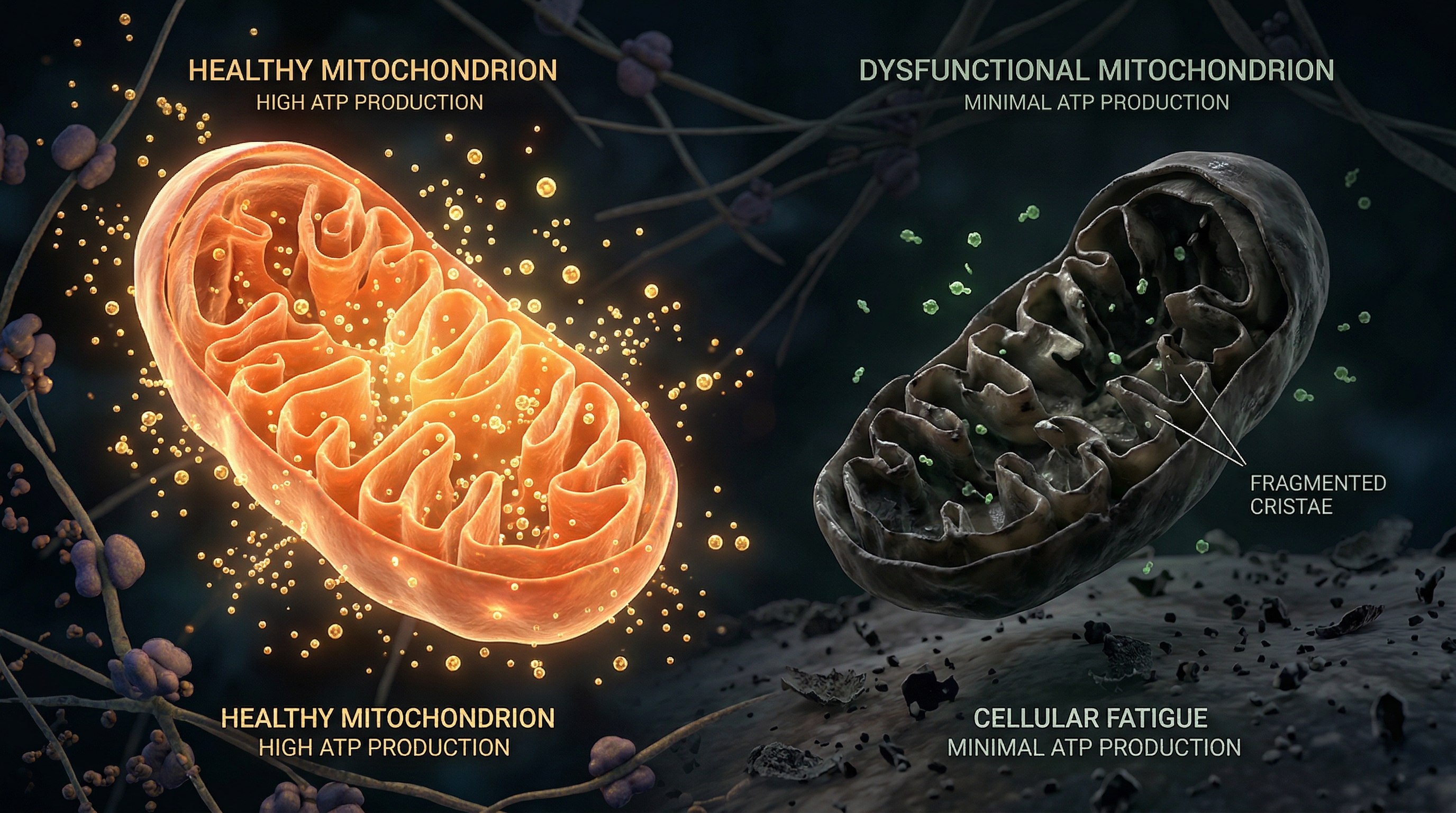
To understand how mitochondrial dysfunction causes fatigue, consider what fatigue actually is at the molecular level. Every movement you make, every thought you think, every heartbeat depends on ATP — the energy currency produced by your mitochondria. When mitochondria can't produce ATP efficiently, your body enters a state of chronic energy deficit.
In a healthy person, mitochondria generate approximately 36 ATP molecules per glucose molecule through oxidative phosphorylation. In ME/CFS patients, multiple studies have found evidence that this process is impaired at several levels:
- Reduced ATP levels: A meta-analysis by Filler et al. (2014) reviewed 18 studies and found consistent evidence of reduced ATP production and elevated lactate in CFS/ME patients — a pattern that indicates cells are shifting toward less efficient anaerobic metabolism to compensate.
- Complex V (ATP synthase) defects: Missailidis et al. (2020) isolated peripheral blood mononuclear cells from ME/CFS patients and found specific inefficiency in Complex V — the molecular turbine that actually synthesizes ATP. This wasn't a general problem with all complexes; it was a targeted failure in the final step of energy production.
- Abnormal exercise response: Tomas et al. (2020) demonstrated that ME/CFS patients' permeabilized cells showed reduced mitochondrial complex activity and abnormal lactate accumulation during exercise. This maps directly onto the hallmark symptom of post-exertional malaise — the devastating worsening of symptoms after physical activity.
The Systematic Evidence: 25 Studies and Counting
In 2020, Holden and colleagues published a landmark systematic review analyzing 25 studies on mitochondrial function in ME/CFS. The consistency of the findings was striking: across different patient populations, measurement techniques, and research groups, the same pattern emerged — reduced mitochondrial membrane potential, impaired oxidative phosphorylation, elevated oxidative stress markers, and structural abnormalities visible on electron microscopy.
This is not a single lab's outlier finding. It's a convergent signal from independent research teams across multiple countries and decades.
A more recent 2025 review by Syed et al. synthesized the growing body of evidence, identifying disruptions in endoplasmic reticulum stress responses, WASF3 signaling (a protein involved in mitochondrial dynamics), and quality control mechanisms like mitophagy — the process by which cells recycle damaged mitochondria.
What Goes Wrong: The Mechanisms
Oxidative Stress and the Vicious Cycle
When mitochondria are damaged, they don't just produce less energy — they produce more reactive oxygen species (ROS). These free radicals damage mitochondrial DNA, membrane lipids, and proteins, further impairing function and generating even more ROS. This self-amplifying cycle can be triggered by viral infections, environmental toxins, or chronic psychological stress — all of which have been identified as risk factors for ME/CFS.
The body has antioxidant defenses (superoxide dismutase, glutathione, catalase), but in chronic mitochondrial dysfunction, these systems can become overwhelmed. Studies consistently show elevated oxidative stress markers in ME/CFS patients, including increased malondialdehyde (a lipid peroxidation product) and reduced total antioxidant capacity.
Mitophagy Failure
Healthy cells constantly monitor their mitochondria, tagging damaged ones for recycling through a process called mitophagy. The PINK1/Parkin pathway acts as a quality control system: when a mitochondrion loses its membrane potential, PINK1 accumulates on its surface, recruits Parkin, and tags the organelle for autophagic degradation.
In ME/CFS, evidence suggests this quality control system may be impaired. Missailidis et al. (2020) found dysregulated mitophagy markers in patient cells, meaning damaged mitochondria may persist in the cell — leaking ROS, producing suboptimal ATP, and consuming cellular resources without contributing useful work.
The Exercise Intolerance Paradox
Perhaps no symptom better illustrates mitochondrial dysfunction in ME/CFS than post-exertional malaise (PEM). In healthy individuals, exercise stimulates mitochondrial biogenesis and improves function. In ME/CFS patients, exercise can trigger a multi-day crash.
A 2025 study by Scheibenbogen et al. used electron microscopy to examine skeletal muscle biopsies from ME/CFS patients and found severely damaged mitochondria — with disrupted cristae (the inner membrane folds where energy production occurs) and fragmented morphology. When these patients exercised, the damage worsened rather than improved, the opposite of what happens in healthy muscle.
This makes biological sense: if your mitochondria are already operating at the edge of their capacity, exercise — which dramatically increases energy demand — can push them past the point of functional failure. The result is an energy crisis at the cellular level that manifests as crushing fatigue, pain, cognitive impairment, and immune dysregulation.
The Genetic Dimension
Not everyone who encounters the same triggers develops ME/CFS. Why? Genetics likely plays a role. Billing-Ross et al. (2016) found that specific mitochondrial DNA variants correlated with symptom severity in ME/CFS patients. Certain mtDNA haplogroups — population-specific genetic variants inherited through the maternal line — may make some individuals more vulnerable to mitochondrial dysfunction under stress.
This genetic predisposition, combined with environmental triggers like viral infection, could explain why ME/CFS often begins after an acute illness. The virus stresses an already marginal mitochondrial system, pushing it past a threshold from which recovery becomes difficult.
What Can Be Done?
The research is clear that ME/CFS involves real, measurable mitochondrial dysfunction. So what interventions show promise?
Coenzyme Q10
CoQ10 is an essential component of the electron transport chain, shuttling electrons between Complexes I/II and Complex III. Multiple studies have found reduced CoQ10 levels in ME/CFS patients, and supplementation has shown benefit in some clinical audits. Myhill et al. (2009, 2013) reported that a combined nutritional protocol targeting oxidative phosphorylation improved ATP profiles in approximately 70% of patients in their clinical practice.
NAD⁺ Precursors
Nicotinamide adenine dinucleotide (NAD⁺) is essential for mitochondrial function, serving as an electron carrier in the Krebs cycle and ETC. NAD⁺ levels decline with age and stress. NMN and NR (nicotinamide riboside) supplementation aims to restore NAD⁺ availability and support sirtuin-mediated mitochondrial quality control.
Pacing, Not Pushing
Perhaps the most important intervention is behavioral: pacing. Because post-exertional malaise reflects genuine cellular energy failure, the traditional advice to "push through" fatigue is counterproductive. Energy management that respects mitochondrial capacity — staying within the "energy envelope" — is now recognized as a cornerstone of ME/CFS management.
Key takeaway: Chronic fatigue is not a failure of willpower. It's a failure of cellular energy production. The research increasingly shows that mitochondrial dysfunction — measurable, specific, and consistent — is a central feature of ME/CFS. Understanding this changes everything: from how we validate patients' experiences to how we design treatments.
References
- Filler K et al. "Association of Mitochondrial Dysfunction and Fatigue: A Review of the Literature." BBA Clinical, 2014;1:12-23.
- Holden S et al. "Systematic Review of Mitochondrial Abnormalities in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome." Journal of Translational Medicine, 2020;18:290.
- Missailidis D et al. "An Isolated Complex V Inefficiency and Dysregulated Mitochondrial Function in Immortalized Lymphocytes from ME/CFS Patients." International Journal of Molecular Sciences, 2020;21(3):1074.
- Tomas C et al. "Cellular Bioenergetics Is Impaired in Patients with Chronic Fatigue Syndrome." PLoS ONE, 2020;15(1):e0227285.
- Tomas C et al. "Mitochondrial Network Parameters in Peripheral Blood Mononuclear Cells Are Associated with Exercise Capacity in ME/CFS." Translational Medicine of Aging, 2019;3:84-93.
- Billing-Ross P et al. "Mitochondrial DNA Variants Correlate with Symptoms in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome." Journal of Translational Medicine, 2016;14:19.
- Sweetman E et al. "SWATH-MS Proteomics Reveals Altered Mitochondrial and Immune Proteins in ME/CFS." Proteomics Clinical Applications, 2020;14(5):e1900151.
- Scheibenbogen C et al. "Skeletal Muscle Biopsy Reveals Severely Damaged Mitochondria in ME/CFS." Annals of Internal Medicine, 2025.
- Syed AA et al. "Mitochondrial Dysfunction in ME/CFS: A Comprehensive Review." Physiological Reviews, 2025.
- Myhill S, Booth NE, McLaren-Howard J. "Chronic Fatigue Syndrome and Mitochondrial Dysfunction." International Journal of Clinical and Experimental Medicine, 2009;2(1):1-16.