The Vagus Nerve and Mitochondria: A Surprising Connection
The Vagus Nerve and Mitochondria: A Surprising Connection
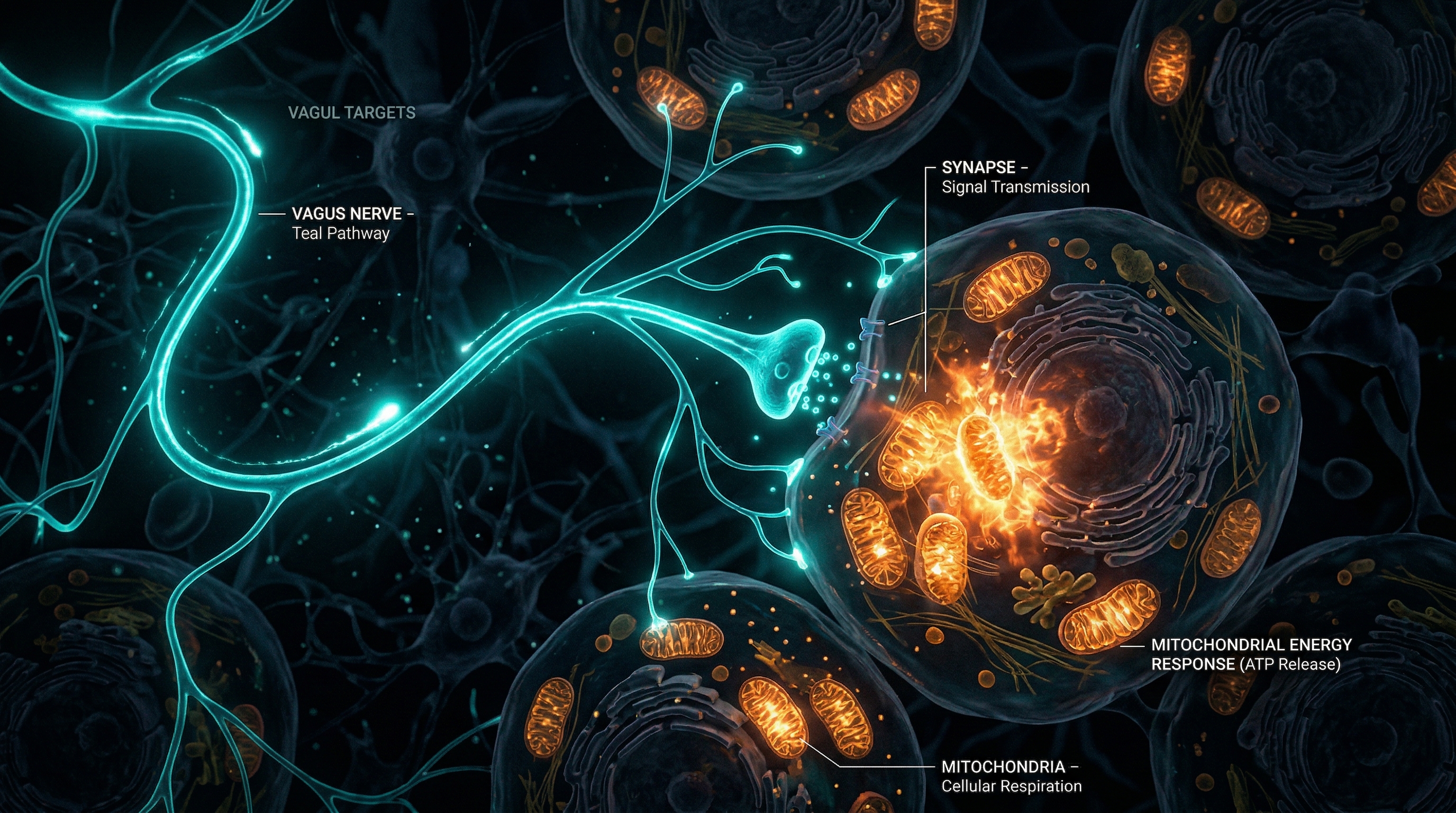
Here's a sentence that might reframe how you think about your body: the health of your mitochondria — those tiny power plants inside every cell — is directly influenced by a single cranial nerve that runs from your brainstem to your gut.
The vagus nerve, the longest of the twelve cranial nerves, has long been recognized as the body's master regulator of parasympathetic function — controlling heart rate, digestion, and respiratory rhythm. But emerging research over the past decade has uncovered something far more profound: the vagus nerve communicates directly with your mitochondria, modulating their function, protecting them from damage, and influencing how efficiently they produce the energy that keeps you alive.
This connection isn't just academically interesting. It has practical implications for anyone dealing with chronic inflammation, fatigue, metabolic dysfunction, or the cellular wear-and-tear of modern life.
The Vagus Nerve: More Than a Relaxation Highway
The vagus nerve (from the Latin vagus, meaning "wandering") is aptly named. It meanders from the medulla oblongata through the neck, chest, and abdomen, branching into the heart, lungs, liver, spleen, pancreas, and intestinal tract. It's roughly 80% afferent (carrying information from the body to the brain) and 20% efferent (carrying instructions from the brain to the body).
This bidirectional traffic is crucial. The vagus nerve isn't just telling your organs to calm down — it's constantly receiving feedback about the state of your tissues, including inflammatory signals, metabolic status, and cellular stress. When this feedback loop is functioning well, your body maintains homeostasis with remarkable efficiency. When it's disrupted, the consequences ripple all the way down to the mitochondria.
The Cholinergic Anti-Inflammatory Pathway
The most well-characterized mechanism linking the vagus nerve to cellular health is the cholinergic anti-inflammatory pathway (CAP), described in detail by researchers at the Feinstein Institute and more recently reviewed in Frontiers in Neuroscience (2024).
Here's how it works:
- Inflammatory detection: When immune cells in the spleen and other organs detect pro-inflammatory signals (bacterial products, cytokines), vagal afferents carry this information to the brainstem.
- Cholinergic response: The brainstem responds by activating vagal efferents, which release acetylcholine (ACh) at nerve terminals near immune cells.
- Receptor binding: Acetylcholine binds to α7 nicotinic acetylcholine receptors (α7nAChR) on macrophages and other immune cells.
- Cytokine suppression: This binding inhibits the NF-κB signaling pathway, dramatically reducing the production of pro-inflammatory cytokines — TNF-α, IL-1β, IL-6, and HMGB1.
The elegance of this system is its precision. Unlike anti-inflammatory drugs that suppress immune function globally, the cholinergic pathway acts locally — dialing down inflammation where it's excessive without compromising the immune system's ability to fight actual threats.
This anti-inflammatory signaling doesn't just calm the immune system — it directly protects mitochondria from the oxidative damage that chronic inflammation causes. When pro-inflammatory cytokines run unchecked, they trigger mitochondrial dysfunction, and dysfunctional mitochondria produce more inflammatory signals — a vicious cycle that the vagus nerve is uniquely positioned to interrupt.
How Vagal Tone Shapes Mitochondrial Function
Mitochondrial Dynamics: Fission and Fusion
Healthy mitochondria are not static structures. They constantly undergo fission (splitting) and fusion (merging) — a dynamic balance that allows them to share components, distribute energy production, and segregate damaged material for repair or disposal.
Chronic inflammation disrupts this balance. Research shows that inflammatory stress upregulates the fission proteins Drp1 and Fis-1 while suppressing the fusion proteins OPA1, Mfn1, and Mfn2. The result: mitochondria fragment into small, inefficient units that produce less ATP and generate more reactive oxygen species (ROS).
Vagus nerve stimulation (VNS) reverses this. Studies in myocardial ischemia models demonstrated that VNS restored the balance between fission and fusion by activating the M3 muscarinic receptor → CaMKKβ → AMPK signaling pathway. This pathway promotes mitochondrial biogenesis (creating new mitochondria), reduces excessive fission, and enhances fusion — effects that were blocked when M3 receptors or AMPK were pharmacologically inhibited.
ATP Production and Membrane Potential
Mitochondria generate ATP through oxidative phosphorylation (OXPHOS) — a process that depends on maintaining a voltage gradient (membrane potential) across their inner membrane. Inflammatory damage disrupts this gradient, causing the electron transport chain to leak electrons (producing superoxide radicals) instead of efficiently generating ATP.
Research in post-cardiac arrest models showed that VNS significantly boosted OXPHOS capacity in hippocampal mitochondria, particularly at Complex I and Complex II of the electron transport chain. Animals treated with VNS showed improved neurological outcomes, likely through enhanced ATP production and reduced oxidative damage.
The Nrf2/HO-1 Antioxidant Defense
Beyond inflammation control, vagal stimulation activates the Nrf2 (nuclear factor erythroid 2-related factor 2) pathway — the master regulator of cellular antioxidant defense. When Nrf2 translocates to the nucleus, it upregulates heme oxygenase-1 (HO-1) and other antioxidant enzymes that scavenge ROS before they can damage mitochondrial DNA, proteins, and membranes.
This means vagal tone doesn't just protect mitochondria from external inflammatory threats — it strengthens their internal defenses against the oxidative byproducts of their own energy production.
The Metabolic Dimension: Glucose and Fat
The vagus nerve's influence on mitochondria extends beyond protection into metabolic optimization. Vagal stimulation enhances AMPK-mediated glucose uptake by increasing Glut4 transporter expression on cell membranes, ensuring that mitochondria receive adequate fuel. Simultaneously, it promotes fatty acid oxidation by upregulating CPT1α — the enzyme that shuttles fatty acids into the mitochondrial matrix for beta-oxidation.
In obese, insulin-resistant animal models, chronic VNS prevented cardiac mitochondrial dysfunction — reducing ROS production, preventing structural damage (swelling, cristae disruption), and preserving ATP-dependent contractile function. These metabolic improvements were accompanied by reduced TNF-α levels and increased adiponectin sensitivity.
The implication is striking: the vagus nerve acts as a metabolic thermostat, tuning mitochondrial function to match the body's energy demands while simultaneously protecting mitochondria from the damage that metabolic overload creates.
The Long COVID Connection
One of the most compelling recent applications of vagus-mitochondria research has emerged from the study of long COVID. Researchers at multiple institutions have proposed that SARS-CoV-2 disrupts a vagal-HPA-mitochondrial axis, creating a self-perpetuating cycle of dysfunction:
- Viral infection damages vagal afferents and efferents
- Impaired vagal signaling reduces cholinergic anti-inflammatory tone
- HPA axis dysregulation leads to cortisol abnormalities
- Without adequate vagal protection, mitochondria suffer oxidative damage, OXPHOS failure, and NLRP3 inflammasome activation
- The result: chronic fatigue, brain fog, exercise intolerance, and persistent inflammation
This framework helps explain why interventions that restore vagal tone — deep breathing exercises, meditation, cold exposure, and non-invasive vagus nerve stimulation devices — show promise in alleviating long COVID symptoms. They're not just "managing stress"; they're reactivating the neural circuitry that protects cellular energy production.
Practical Ways to Support Your Vagus-Mitochondria Axis
You don't need a medical device to improve your vagal tone. Several accessible practices have been shown to enhance vagal signaling:
- Slow, diaphragmatic breathing: Breathing at approximately 6 breaths per minute (the "resonance frequency") maximizes heart rate variability — a direct marker of vagal tone.
- Cold exposure: Even brief cold water exposure (30–60 seconds) activates the vagus nerve through cold receptors in the face and neck.
- Singing and humming: The vagus nerve innervates the vocal cords. Vibrations from singing, chanting, or humming stimulate vagal afferents.
- Meditation and mindfulness: Regular practice is associated with improved heart rate variability and reduced inflammatory markers.
- Exercise: Moderate aerobic exercise enhances vagal tone and has direct benefits for mitochondrial biogenesis through AMPK activation.
- Gut health: The vagus nerve communicates with the gut microbiome. A healthy, diverse microbiome sends positive signals back through vagal afferents.
The Emerging Frontier
Researchers are now exploring whether targeted vagus nerve stimulation — using non-invasive devices that deliver electrical or acoustic energy to the auricular branch of the vagus — can be used therapeutically to restore mitochondrial function in specific diseases. Early clinical trials in rheumatoid arthritis and inflammatory bowel disease have shown reduced CRP levels and symptom improvement. Trials targeting metabolic syndrome and neurodegenerative diseases are underway.
The vagus nerve, it turns out, is not just the body's relaxation switch. It's a sophisticated communication network that reaches into the most fundamental machinery of life — the mitochondria — and tells them how to behave. Understanding this connection gives us a new lever for health: not just managing symptoms, but tuning the cellular machinery that determines whether symptoms arise in the first place.
References
- Frontiers in Neuroscience (2024). "Vagus nerve stimulation and the cholinergic anti-inflammatory pathway." doi: 10.3389/fnins.2024.1490300
- PMC11671747. "The vagus-HPA-mitochondrial axis in long COVID."
- PMC5192749. "VNS restores mitochondrial dynamics via M3R/CaMKKβ/AMPK pathway in myocardial ischemia."
- Frontiers in Neuroscience (2022). "Vagus nerve stimulation enhances hippocampal OXPHOS capacity." doi: 10.3389/fnins.2022.762007
- Nature Scientific Reports. "Chronic VNS prevents cardiac mitochondrial dysfunction in obesity." doi: 10.1038/srep19749
- Pavlov VA, Tracey KJ. "The cholinergic anti-inflammatory pathway." Brain, Behavior, and Immunity.
- Howland RH. "Vagus nerve stimulation." Current Behavioral Neuroscience Reports.